What is the Triple Point?
Most of us learn early that water exists in three forms — solid, liquid, and gas — and that temperature determines which state we get. But this is only half the story. Pressure matters just as much as temperature. And at one very specific combination of both, something extraordinary happens: all three phases coexist in perfect equilibrium, simultaneously, in the same container. This is the triple point.
The triple point of water sits at 273.16 K (0.01 °C) and a pressure of 611.657 Pa (0.00604 atm) — far below the atmospheric pressure we live under. At this precise coordinate on the pressure-temperature diagram, the substance neither fully melts, boils, nor freezes. Ice, liquid water, and water vapor exist side by side indefinitely.
The Phase Diagram
To understand the triple point, it helps to picture a phase diagram — a graph of pressure on the vertical axis and temperature on the horizontal axis. Every combination of pressure and temperature corresponds to a single stable phase (or a boundary between phases).
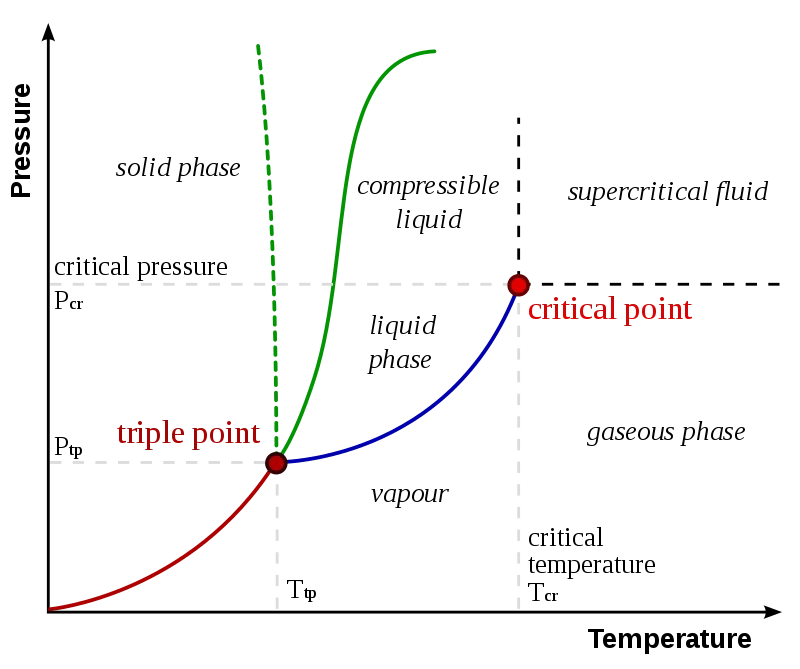
Three curves meet at the triple point:
- The solid–liquid boundary (the melting curve): crossing this means going between ice and water
- The liquid–gas boundary (the vaporization curve): crossing this means water boiling or condensing
- The solid–gas boundary (the sublimation curve): crossing this means ice turning directly to vapor, or vapor depositing directly as ice — no liquid phase at all
At the triple point, all three curves intersect. There is no "solid side" or "liquid side" — all three coexist.
Why Can't This Happen at Normal Pressure?
At sea level, we never see ice, water, and steam in equilibrium because the atmosphere's pressure (101,325 Pa) forces water through a specific sequence: it melts at 0 °C and boils at 100 °C. To reach the triple point, we must lower the pressure dramatically — to just 0.006 atm. This is why the triple point only exists in a sealed, evacuated environment.
The underlying reason is thermodynamic: at the triple point, the chemical potential of all three phases is equal. Chemical potential is a measure of how much a molecule "wants" to be in a given phase. When all three potentials match exactly, there is no thermodynamic preference — no net driving force to convert any one phase into another. The system is in complete, stable equilibrium.
The Ideal Gas Equation and Boiling Points
The connection to pressure can be seen through the ideal gas equation:
For a liquid in equilibrium with its vapor, the Clausius-Clapeyron equation describes how the boiling point shifts with pressure:
where is the latent heat of vaporization and is the volume change between liquid and gas. As pressure decreases, the boiling point drops — steeply at first, then approaching the triple point. Below the triple-point pressure, liquid cannot exist at all: the substance either freezes or sublimes.
The Triple Points of Different Substances
Every substance has its own triple point. Here is a selection:
| Substance | Temperature | Pressure |
|---|---|---|
| Hydrogen | 13.84 K | 7.04 kPa |
| Neon | 24.56 K | 43.2 kPa |
| Oxygen | 54.36 K | 0.15 kPa |
| Argon | 83.81 K | 68.9 kPa |
| Mercury | 234.31 K | 0.000165 Pa |
| Water | 273.16 K | 0.6117 kPa |
| Ethylene carbonate | 309.47 K | — |
| Benzoic acid | 395.48 K | — |
The Critical Point
Following the liquid–gas boundary upward in pressure and temperature, it doesn't go on forever. It ends at the critical point — for water, 647.1 K and 22.06 MPa. Above the critical point, the distinction between liquid and gas disappears entirely, and the substance becomes a supercritical fluid: a single continuous phase with properties of both liquid and gas simultaneously.
Practical Applications: Metrology
The triple point's most important application is in precision thermometry. Because it occurs at an exactly reproducible temperature for a pure substance, it serves as a fixed calibration point.
The International Temperature Scale of 1990 (ITS-90) — the global standard for temperature measurement — relies on the triple points of:
- Hydrogen (13.8033 K)
- Neon (24.5561 K)
- Oxygen (54.3584 K)
- Argon (83.8058 K)
- Mercury (234.3156 K)
- Water (273.16 K, the defining fixed point)
Special triple-point cells — sealed glass vessels containing ultra-pure substances — are used in national metrology laboratories worldwide. A platinum resistance thermometer placed in such a cell, maintained at the triple point, is calibrated to an absolute accuracy of ±0.0001 K.
Substances with higher triple-point temperatures are also useful for practical calibration. Ethylene carbonate (36.315 °C) is used to calibrate clinical thermometers. Benzoic acid (122.33 °C) calibrates thermometers used in sterilization equipment.
Freeze-Drying: Triple Point in Action
One of the most common industrial applications of the triple-point concept is freeze-drying (lyophilization). Food or pharmaceuticals are first frozen solid, then placed in a vacuum chamber. When pressure is reduced below the triple-point pressure of water (0.006 atm), ice sublimes directly into vapor without ever passing through the liquid phase. This removes moisture while preserving structure, flavor, and biological activity — the reason astronaut food and instant coffee exist.
The triple point is not just a curiosity — it is the most precisely reproducible physical state in nature. A substance's triple-point temperature is as fundamental as a physical constant, independent of any external reference. This is why the kelvin itself was redefined in 1954 to be anchored to the triple point of water.