The Anomalous Expansion of Water
Among all the substances in the universe, water is one of the strangest. While almost every other liquid contracts continuously as it cools — becoming denser and denser right up to the freezing point — water breaks this rule in a way that has profound consequences for life on Earth. Between 4 °C and 0 °C, water actually expands as it gets colder, becoming less dense as it approaches its freezing point.
This is the anomalous expansion of water, and understanding it requires going all the way down to the structure of individual molecules.
The Normal Behavior of Liquids
When you cool most liquids, the molecules slow down, vibrate less, and pack together more tightly. The result is a steady, predictable increase in density as temperature drops. When the liquid freezes, it contracts further — the solid is denser than the liquid, and it sinks.
Water follows this pattern from 100 °C down to about 4 °C, behaving exactly as expected. But below 4 °C, the density curve reverses. And at 0 °C, water freezes into ice — which, famously, floats.
The Molecular Origin: Hydrogen Bonding
The key is the shape of the water molecule and the unique bonds it forms.
A water molecule () has a bent geometry: the oxygen atom sits at the center, with two hydrogen atoms attached at an angle of about 104.5°. Oxygen is strongly electronegative — it pulls the shared electrons toward itself, leaving the hydrogen ends with a partial positive charge () and the oxygen end with a partial negative charge (). This makes water a polar molecule.
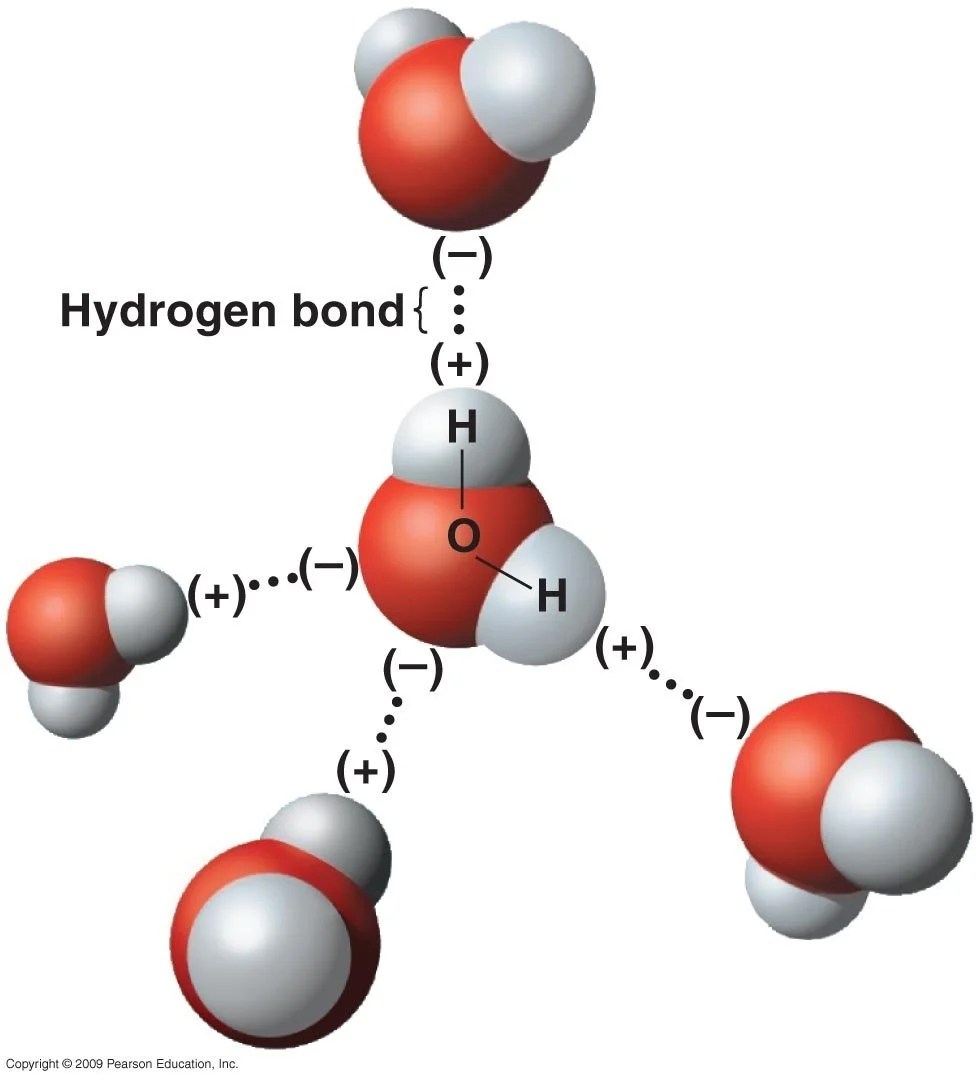
Because of this polarity, the positive end of one water molecule attracts the negative end of a neighbor. This electrostatic attraction is called a hydrogen bond — weaker than a covalent bond, but strong enough to significantly structure the liquid.
At temperatures above 4 °C, thermal energy keeps the molecules in a largely disordered arrangement. The molecules are close together because hydrogen bonds are constantly forming and breaking, but the geometry isn't particularly open.
What Happens Below 4 °C
As water cools toward 4 °C and below, thermal motion decreases enough that hydrogen bonds become more persistent. The molecules begin organizing into a tetrahedral lattice structure — each water molecule hydrogen-bonds to four neighbors arranged at the corners of a tetrahedron.
This structure is more open than the disordered arrangement in warmer liquid water. The tetrahedral geometry requires more space between molecules than random thermal jostling allows. As a result, the volume increases even as the temperature drops — the density falls.
At 0 °C, this lattice crystallizes fully into ice Ih (ordinary hexagonal ice), with each molecule locked into the tetrahedral framework. Ice is about 9% less dense than liquid water at 4 °C — which is why ice floats.
The Density Curve
The density of water peaks at exactly 3.98 °C (often rounded to 4 °C), reaching approximately 999.97 kg/m³ — its maximum. The curve is remarkably sharp near the peak:
| Temperature | Density (kg/m³) |
|---|---|
| 10 °C | 999.70 |
| 4 °C | 999.97 (maximum) |
| 0 °C (liquid) | 999.84 |
| 0 °C (ice) | 917.0 |
The expansion between 4 °C and 0 °C is subtle — less than 0.1% for liquid water — but the jump to ice is dramatic.
Why Water Freezes From the Top Down
The anomalous expansion is the reason lakes, rivers, and ponds freeze from the surface downward rather than from the bottom up.
Consider a lake cooling in autumn:
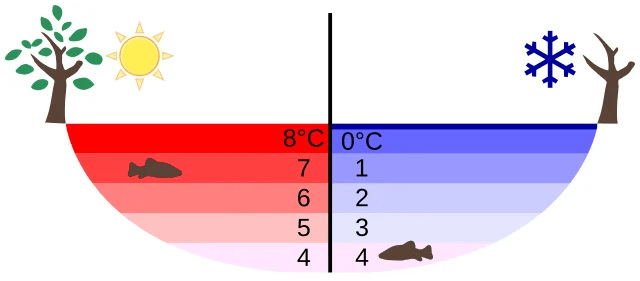
- Surface water cools from, say, 20 °C down to 4 °C. As it cools, it becomes denser and sinks, displacing warmer water upward. This convective mixing continues until the entire lake reaches 4 °C — its maximum density.
- Once the whole lake is at 4 °C, further cooling makes the surface water less dense. It no longer sinks. Instead, it stays at the top and continues cooling.
- The surface reaches 0 °C and freezes. Ice forms a layer on top.
- Ice acts as an insulating blanket. Its low thermal conductivity slows further heat loss. The water below remains liquid, hovering near 4 °C.
This means that even in the harshest winters, the lake bottom remains liquid — and aquatic life survives.
If water behaved normally (denser when colder, all the way to freezing), lakes would freeze from the bottom up. Ice would form on the floor and work its way upward, potentially freezing the entire lake solid and killing everything inside.
Consequences for the Earth's Climate
The anomalous expansion of water has effects beyond individual lakes:
- Ocean thermohaline circulation: The density maximum at 4 °C influences how ocean water circulates. Dense, cold water sinks at the poles and drives global ocean currents.
- Weathering and erosion: Water that seeps into rock cracks expands when it freezes, exerting pressures of up to 2,000 atm — enough to shatter granite. This freeze-thaw cycle is one of the primary forces shaping mountains and landscapes over geological timescales.
- Biological antifreeze: Many organisms in cold environments have evolved antifreeze proteins to prevent ice crystals from forming in their cells — a direct evolutionary response to the fact that water expands and ice crystals are structurally damaging.
Burst Pipes: The Engineering Problem
The ~9% expansion of water as it freezes generates enormous pressure inside confined spaces. In a sealed pipe, freezing water can exert pressures of over 100 MPa (1,000 atm). Most pipe materials — copper, iron, even some plastics — cannot withstand this force.
This is why pipes burst in freezing weather: it's not the ice itself cracking the pipe, but the water pressure that builds up between the advancing ice front and a closed valve or fitting. The solution — insulating pipes, allowing a slow drip to keep water moving, or using flexible tubing — all work by preventing that pressure from building up.
Water's anomalous expansion is not a minor footnote in chemistry — it is a planetary life-support system. Without it, lakes would freeze solid each winter, aquatic ecosystems would collapse, and the cycling of heat through Earth's oceans would operate completely differently. The hydrogen bond, a force barely a twentieth the strength of a covalent bond, turns out to be one of the most consequential forces in the biosphere.